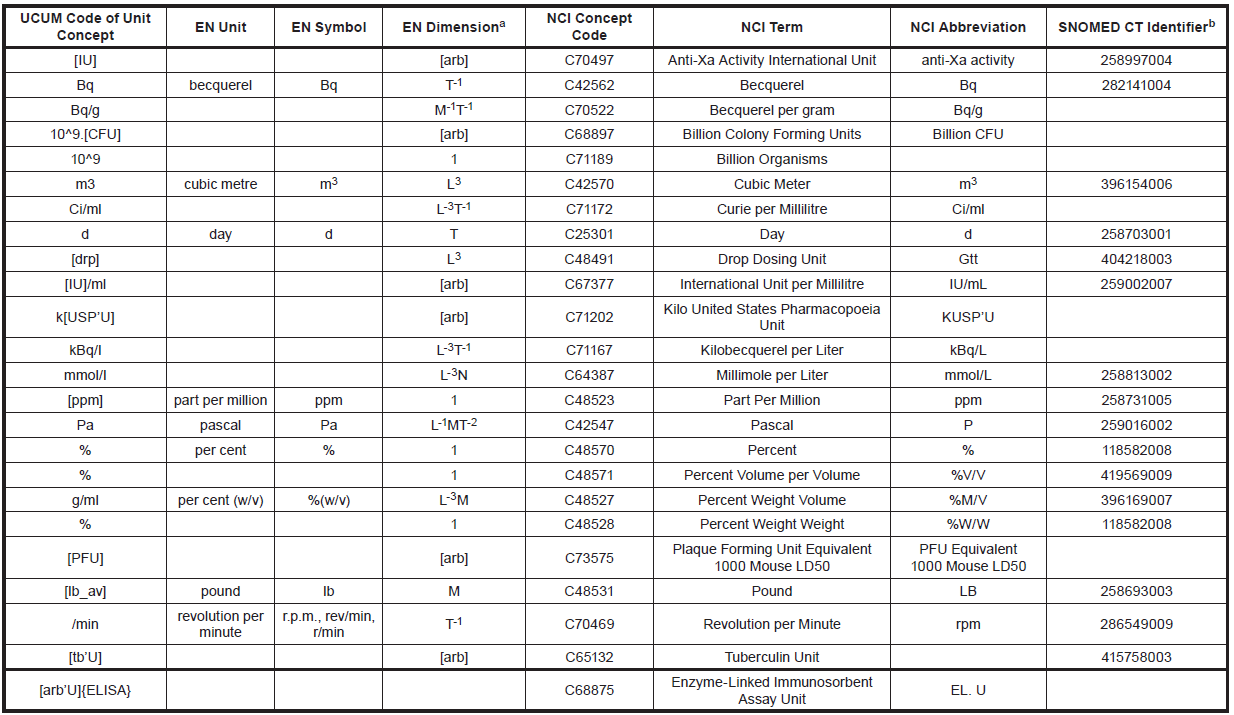
ISO 11240 – Units of Measurement
What ISO 11240 does:
- specifies rules for the usage of units of measurement for IDMP
- specifies requirements for traceability to metrological standards
- establish reference code system for units
- provide structures and rules for mapping between different unit vocabularies and language translations
- linking to existing systems, dictionaries, and repositories
Strength of a medicinal product based on ISO 11240
In the case of strength given on a per dose basis, additional information on the characteristics of the countable dose item
may be required when comparing to strengths of other products.
EXAMPLE 1 Expression “per dose”: Total amount of substance per total amount of solution (200 mg of substance
contained in 10 ml of solution), total mass of ingredient contained in one single dose (500 mg of substance contained in
one tablet). Note that the description of the component (substance, solution, tablet) is not part of the unit of measurement.
EXAMPLE 2 Expression as intensive quantity: Mass concentration 20 mg/ml, activity content 5,000 IU/g.
EXAMPLE 3 Using the unit symbol “U”
The unit “U” of enzymatic activity was defined in 1964 by the International Union of Biochemistry as the
catalytic activity that catalyses the transformation of 1 µmol of the substrate per minute. Typical biological
enzyme activities are in the range of 1 U to 100 U. This unit of measurement can also be expressed explicitly
as “µmol/min”, where enzymatic activity is the property being addressed. Still, some arbitrariness is involved in
the definition of “standard conditions” (e.g. temperature, pH).
This unit denoted by the symbol “U” is not an arbitrary unit. In order to ensure traceability and avoid ambiguities,
it is important to discriminate this meaning against the frequently encountered usage of the same symbol “U”
for other (arbitrary) units. This is especially required to ensure unique meaning of codes used for information
exchange. “U” is assigned to enzymatic activity in the UCUM code system. However, the following description
uses the symbol “U” for a completely unrelated concept. Here “U” stands for an arbitrary unit that is defined
based on a “bioassay”:
e.g. Concentration 5 000 U/ml solution for injection.